|
Perhaps in part that's because Smithson Tennant, an English chemist, was also the first to establish that diamond is a form of carbon.so this was never going to match up to that glittering career highlight. Even the man who discovered osmium treated it rather sniffily. It's brittle, prone to ponginess and arguably the dunce of the periodic table. Under-appreciated under-exploited osmium - if any element needs a change of PR this is the one. Given the whole periodic table to choose from, how to pick a particular element to talk about rather than any other? They've all got their charms and quirks - well, except maybe bismuth.I've never had much time for bismuth - but the deal was I had to single out one. Political stability of top reserve holderĪ percentile rank for the political stability of the country with the largest reserves, derived from World Bank governance indicators. The higher the value, the larger risk there is to supply.Ī percentile rank for the political stability of the top producing country, derived from World Bank governance indicators. The percentage of the world reserves located in the country with the largest reserves. The higher the value, the larger risk there is to supply. The percentage of an element produced in the top producing country. Low = substitution is possible with little or no economic and/or performance impact Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. 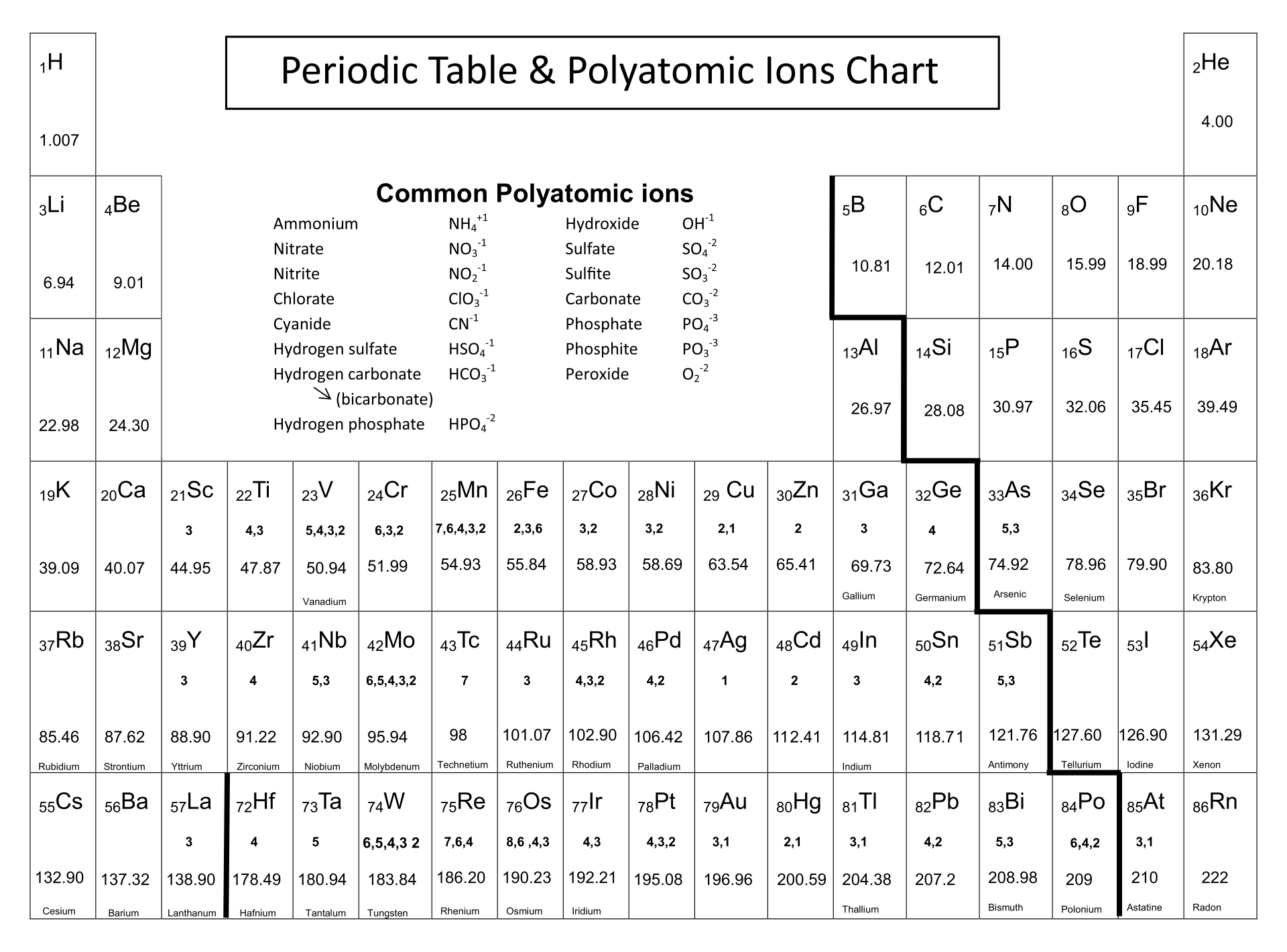
This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). 
The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. F gains 1 e - and Ogains 2 e -'s, and O gain e -'s to attain theconfiguration of Ne thus, the halogens tend to -1 charges and theoxygen family tends to a -2 charge.A vertical column in the periodic table. On the other side of the periodic table elements gain electrons toresemble the next higher noble gas. Thus, the group 2 metals tend tohave a +2 charge. The alkali earth metals (group 2), such as, Mg or Sr lose two e-'sto attain the configuration of Ne. Since K loses one electron (1 negative charge)it is no longer neutral it now has a +1 charge. Kloses an electron so that it will have the same electronconfiguration as Ne. The alkali metals will lose anelectron to resemble the next lowest noble gas thus, all the alkalimetals form +1 ions. Two (or more elements) can share electrons so that each attains the electron configuration of the nearest noble gas. 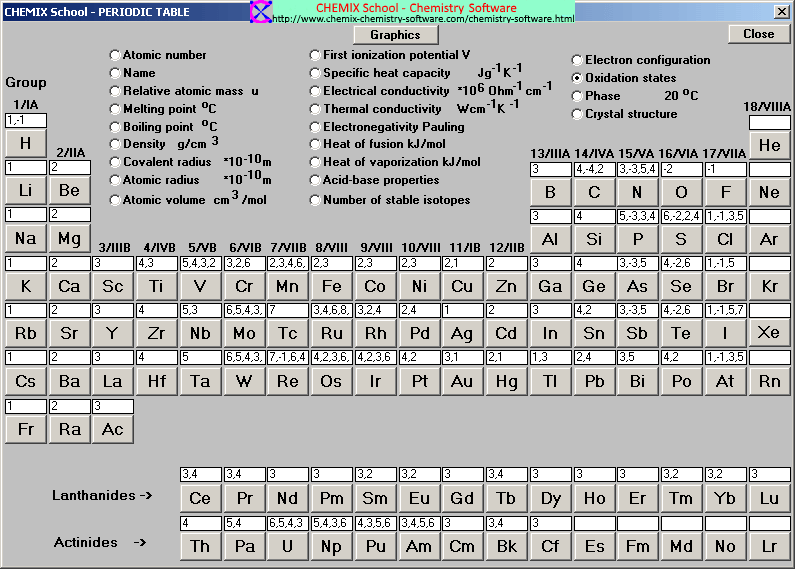
An element can attract additional electrons to achieve the electron configuration of the next highest noble gas.An element can give up electrons to achieve the electron configuration of the next lowest noble gas.Achieving a Noble Gas Electron Configurationįor reasons we will discuss later, elements react until theelement achieves an electron configuration of a noble gas.Įlements can achieve a noble gas electron configuration threeways.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
